CodeĪs can be seen from the comparison table above, SRDX ranks number four in this group of eight stocks with no last dividend. If two stocks are in the same industry and have a similar market capitalisation, then they are considered similar. Stock Price and Quote TIP: Upgrade to FINVIZElite to get real-time quotes, intraday charts, and advanced charting tools. The catheter, which received the FDAs 510(k) clearance in June 2021, is the latest. Surmodics (Nasdaq:SRDX) announced today that it received formal feedback from the FDA related to its SurVeil drug-coated balloon (DCB). 018 RX PTA (percutaneous transluminal angioplasty) Dilatation Catheter. Compared with other stocksĪnother way to look at a stock’s dividend is to compare it to a peer company’s dividend. 018 RX PTA Dilatation Catheters are indicated for Percutaneous Transluminal Angioplasty (PTA). Ancora Heart, Egnite partner on AccuCinch trial enrollment. However there is no dividend record for this stock so far. 
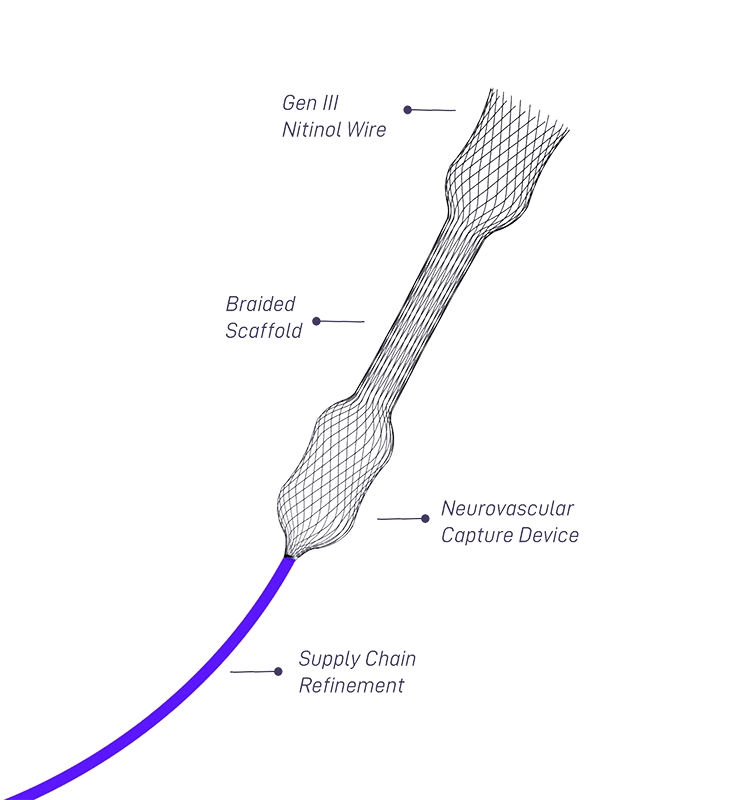
1.10.1 Design Verification (Bench-Top Testing) The physical and mechanical properties of the ACE 68 Reperfusion Catheter were assessed using standard test methods and pre-determined acceptance criteria. We believe using more recent dividend history can provide better dividend analysis for a company. The subject ACE 68 Reperfusion Catheters met all established requirements. For more information about this company, please visit their website. The company was founded in 1979 and is headquartered in Eden Prairie, Minnesota. This segment offers protein stabilization reagents, substrates, surface coatings, and antigens. The IVD segment designs, develops, and manufactures component products and technologies for diagnostic immunoassay, as well as molecular test and biomedical research applications. (NASDAQ:NASDAQ:SRDX) Q2 2022 Earnings Conference Call Ap8:30 AM ETCompany ParticipantsTim Arens - SVP, Finance & CFOGary Maharaj - President &. The Medical Device segment engages in the provision of surface modification coating technologies to enhance access, deliverability, and predictable deployment of medical devices and drug-delivery coating technologies to provide site-specific drug-delivery from the surface of a medical device for coronary, peripheral, neuro-vascular and structural heart, and other markets, as well as design, development, and manufacturing of interventional medical devices, primarily balloons and catheters, including drug-coated balloons for peripheral arterial disease treatment and other applications. The company offers a wide range of advanced balloon catheter, thrombectomy, surface modification coatings and ultra-thin-walled catheter technologies to advance new medical devices, biomedical products, and enhance existing devices. It operates in two segments, Medical Device and In Vitro Diagnostics (IVD). Surmodics, Inc., together with its subsidiaries, provides surface modification technologies for intravascular medical devices, and chemical components for in vitro diagnostic immunoassay tests and microarrays in the United States and internationally.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
